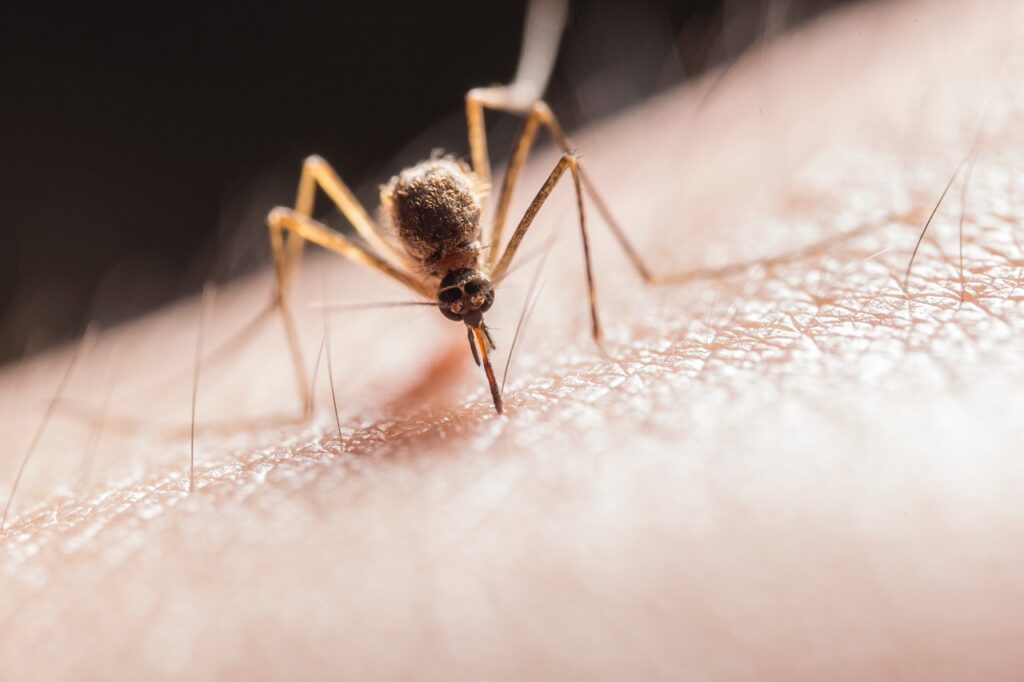
Australian regulators last week approved a new malaria treatment for children that includes a single-dose medication and promises a new weapon in defeating the deadly illness, according to reports.
The Australian Therapeutic Goods Administration (TGA) approved the novel treatment in children aged 2 years and above after accepting the application from GlaxoSmithKline (GSK) and the Medicines for Malaria Venture (MMV) in January.
Both the U.S. Food and Drug Administration (FDA) and TGA approved Kozenis (tafenoquine) for people aged 16 years and older in July 2018, according to GSK. This new approval will help children, who are more likely to die from malaria than adults.
GSK and MMV said they sought approval “to extend the indication of single-dose Kozenis (tafenoquine) to paediatric populations for the radical cure (prevention of relapse) of Plasmodium vivax (P. vivax) malaria.”
GSK said that the application includes “a novel, 50mg dispersible tablet that can be dispersed in water to facilitate use in children.”
According to data from WHO’s 2021 World malaria report, P. vivax malaria causes “between 4.1 and 5.1 million clinical infections every year, and poses a disproportionate burden for children aged 2 to 6 years who are four times as likely as adults to be infected.”
A single dose of tafenoquine was administered along with traditional chloroquine treatment in order to offset the first symptoms, which include fevers, headaches, and chills, and also targets the disease in the liver to prevent a recurrence, GSK said.
A ‘radical cure’
“Most treatments, including chloroquine, are directed at the blood stage of the parasite, and so cannot prevent recurrence of the infection and its associated symptoms. But tafenoquine goes after the sleeper colonies in the liver. In combination with chloroquine, tafenoquine can deliver what scientists call a ‘radical cure,’ ” The New York Times reported.
Thousands of children die from malaria every year, with the majority of those deaths being among children under the age of 5, especially in sub-Saharan Africa, according to The New York Times.
Medicines for Malaria Venture (MMV), a nonprofit that helped to develop the drug, said “the submission was supported by a Phase 2b clinical study (TEACH) that evaluated dosages of tafenoquine based on weight for children between the age of 2 years, and weighing at least 10 kg, and up to 15 years.”
MMV’s Chief Executive Officer, Dr. David Reddy, stressed that “P. vivax malaria is particularly dangerous for young children for whom repeated relapses can lead to cumulative severe anaemia and, in some cases, be fatal.”
Kozenis (tafenoquine) will now be submitted for approval in nine countries where malaria is endemic, as well as the World Health Organization, The New York Times said.