The field of bleeding disorders has seen monumental transformation. For many patients, biotech innovation has turned what was once a fatal diagnosis with a severely shortened life expectancy into a largely manageable chronic condition with a near-normal lifespan. Yet, misconceptions are still common, and many of the millions of patients that live with one around 50 types of bleeding disorders are counting on continued progress.
In observation of March’s National Bleeding Disorder Month, Bio.News sat down with Pathway to Cures—the National Bleeding Disorders Foundation’s donor-based, non-profit venture philanthropy fund created specifically to accelerate the development of treatments and cures for bleeding disorders—to discuss bleeding disorders, their history of treatment, and what is being done to support further innovation for treatments and cures.
What are bleeding disorders? And what do most people get wrong about them?
Bleeding disorders are a group of conditions that impair the body’s ability to form blood clots and control bleeding. The most recognized are hemophilia A and B—inherited deficiencies of clotting factors VIII and IX respectively—as well as VWD, the most common inherited bleeding disorder in the world, affecting roughly 1 in 100 people.
Due to their rarity, bleeding disorders are poorly understood outside the patient community. Perhaps the most pervasive misconception is that hemophilia A and B affect only men. While hemophilia is X-linked and more commonly diagnosed in males, women—often dismissed as “carriers”—can also have hemophilia. Von Willebrand disease affects all sexes equally—and women and girls with bleeding disorders are dramatically underdiagnosed. Heavy menstrual bleeding, for instance, is frequently discounted as “normal” rather than recognized as a potential symptom of an underlying clotting deficiency.
Another widespread myth is that bleeding disorders are uniformly severe. In reality, they span a broad spectrum. Many people live with mild or moderate forms of the condition and may not receive a diagnosis until a dental procedure, surgery, or injury reveals an unexpected bleeding challenge. This diagnostic gap means that patients can go years—sometimes decades—without appropriate care.
Finally, bleeding disorders are not just about bruising or bleeding that won’t stop. They carry invisible burdens: chronic joint damage from internal bleeds, anxiety about daily activities, and the cognitive and logistical weight of managing a complex treatment regimen.
While hemophilia A and B and von Willebrand disease are the most widely known bleeding disorders, there are lesser-known conditions that affect people around the world that may have no therapy available. These include rare factor deficiencies and platelet disorders.
What therapies exist today, and what is the next frontier?
Up until the mid-1960s, the only treatment for hemophilia was whole blood transfusions, and many of those with severe hemophilia lived to be about 20 years of age. In the early 1970s, methods were developed to highly concentrate plasma, enabling people with hemophilia to use factor concentrates to self-treat at home for the first time—opening the way for safer surgeries, reducing dependence on emergency rooms and dramatically improving quality of life. Unfortunately, during the 1980s, the pooled plasma used to make the factor concentrates was contaminated with HIV and hepatitis C, and over half of those with hemophilia using these products were infected. Thousands of people died.
Purification methods were later improved to kill these and other viruses for plasma derived products and synthetic clotting factors. Developed in the 1990s, they also eliminated blood-borne transmission risks and remain a key standard of care today.
Extended half-life (EHL) clotting factors represented the next wave of innovation, reducing the frequency of infusions from every other day to once or twice a week—a meaningful quality-of-life improvement. More recently, non-factor therapies have transformed care for hemophilia A patients. These therapies can be administered subcutaneously as infrequently as once monthly. For a community accustomed to intravenous infusions multiple times per week, this shift has been life-changing.
Innovation in treatment has also resulted in gene therapy for the treatment of hemophilia. Gene therapy seeks sustained or permanent production of the deficient clotting factor potentially freeing patients from a lifetime of prophylactic intravenous infusions. Gene therapy products for hemophilia A and B have received regulatory approval, or are in late-stage trials, and early data show meaningful and durable factor level increases in many patients. Unfortunately, two gene therapies have been pulled off the market. Although they represented major scientific advancement, access was difficult given the one-time high cost, insurance and reimbursement challenges, patient qualification and interest, clinical monitoring requirements, and uncertainty about the durability all influenced decision-making and approval by payers.
Beyond gene therapy, the development pipeline includes RNA-based approaches, engineered protein production, gene editing, next-generation subcutaneous factor replacements, and rebalancing agents that address the coagulation cascade from new angles that could be redosable and have durability of several months before redosing. The field is experiencing great scientific innovation—but translating that science into accessible, affordable treatments for all patients remains an enormous challenge.
Why was Pathway to Cures founded, and how does venture philanthropy work?
Pathway to Cures was founded by the National Bleeding Disorders Foundation (NBDF) as a donor-based non-profit venture philanthropy fund to maximize impact and invest in companies seeking to develop therapies and cures for inheritable blood and bleeding disorders. Donations made to Pathway to Cures are invested in early-stage companies developing therapeutics, diagnostics, and devices to address unmet medical needs. These investments support commercialization of products for the inheritable blood and bleeding disorders community and provide returns to Pathway to Cures. The returns are reinvested, amplifying impact and creating a sustainable funding program to support future investments.
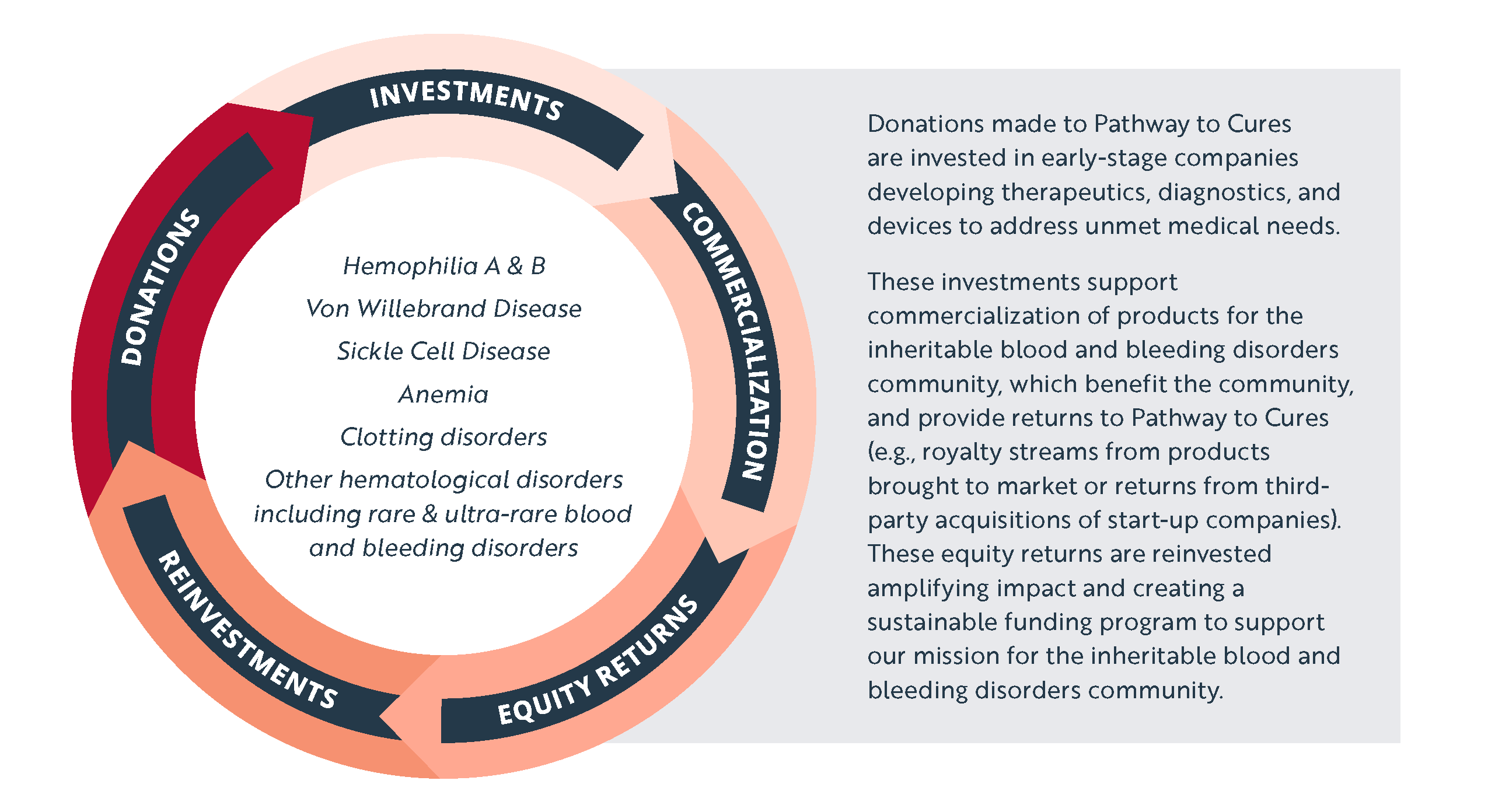 Government grants and academic research are essential, but they often operate on long timelines and with risk profiles that do not favor early-stage, high-risk therapeutic development. Meanwhile, commercial venture capital is driven by return on investment—and rare disease markets, while deeply important to patients, do not always attract private capital at the scale needed.
Government grants and academic research are essential, but they often operate on long timelines and with risk profiles that do not favor early-stage, high-risk therapeutic development. Meanwhile, commercial venture capital is driven by return on investment—and rare disease markets, while deeply important to patients, do not always attract private capital at the scale needed.
Venture philanthropy bridges that gap and puts funding into de-risk opportunities promoting further investment. Pathway to Cures’ leadership apply the tools and discipline of venture capital—milestone-driven funding, active partnership, portfolio thinking—to philanthropic goals. Rather than simply making grants, a venture philanthropy organization like Pathway to Cures invests capital in biotech companies with promising science, takes an equity stake or royalty interest, and deploys hands-on strategic support alongside the funding.
When those investments succeed and generate returns, the proceeds are recycled back into the mission—funding the next round of research. It is a virtuous cycle that allows philanthropic dollars to work harder and go further. Critically, it also keeps the patient at the center: investment decisions are guided not just by commercial potential but by the unmet needs of the bleeding disorders community.
How do you partner with biotech startups, patient groups, and others to advance therapies?
Partnering is at the heart of everything we do. The Pathway to Cures functions as a connector between multiple stakeholders.
With biotech startups, engagement begins early in the scientific development process—often at the preclinical stage, when companies have compelling data but need both capital and strategic guidance to advance toward clinical trials. Pathway to Cures provides funding alongside access to its network of scientific advisors, clinical experts, and regulatory strategists, helping companies navigate the complex path from discovery to approval.
Pathway to Cures’ parent organization, the National Bleeding Disorders Foundation, has been involved in the bleeding disorders community and has a long history (over 75 years) of advocacy, education, and research with deep relationships with patients and families. Pathway to Cures works to align its investment strategy with the lived experience experts (LEEs) to understand unmet needs, ensuring that what gets funded reflects what patients actually need most. LEEs also provide input to leading industry and biotech organizations to ensure the authentic voices of patients are integrated into every stage of clinical development. The earlier companies incorporate patient perspectives, the more effectively therapies can meet real-world needs. At the earliest stages, input from LEEs can shape study design and product development. Research shows that incorporating patient perspectives into clinical trials can improve study design, focus on outcomes that matter most to patients, and increase participant enrollment. (CDER) (BMJ) Conversely, therapies that reach the market without this input risk poor adoption.
What are you most hopeful about this Bleeding Disorders Awareness Month?
This Bleeding Disorders Awareness Month, the focus turns to visibility—for the millions of people living with undiagnosed or undertreated conditions, for the women and girls whose symptoms have been dismissed, and for the communities in lower-resource settings around the world who still lack access to therapies. The goal is to work towards better research and treatments and celebrate the resiliency of those living with a bleeding disorder and help NBDF and Pathway to Cures to live in a world where every person with a bleeding disorder has access to safe, effective, and curative treatment.




