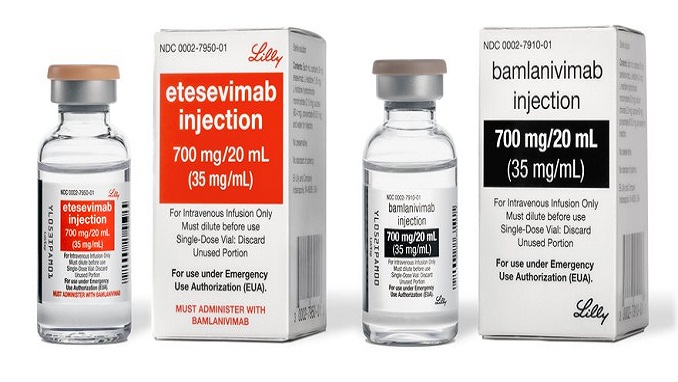
The U.S. Food and Drug Administration last week issued emergency authorization of Eli Lilly’s monoclonal antibody drug in children under the age of 12, including newborn babies.
Eli Lilly’s drug, a mixture of the monoclonal antibodies bamlanivimab and etesevimab, is given to patients who have COVID-19, or have been exposed to the virus, to prevent them from becoming severely ill. “Monoclonal antibodies are laboratory-made proteins that mimic the immune system’s ability to fight off harmful pathogens, such as viruses,” according to the FDA.
The drug received emergency use authorization (EUA) for adults in February and, as of Monday, it is the only monoclonal antibody treatment that can be used for all age groups, including newborns. The EUA designation means the drug is not fully approved by the FDA but can be used in serious, and life-threatening conditions, which includes the risk of COVID-19 infection.
“Now all patients at high risk of severe COVID-19, including children and newborn babies, have an option for treatment and post-exposure prevention,” said Patrizia Cavazzoni, director of the FDA’s Center for Drug Evaluation and Research.
The antibody drugs, which are only administered via injection or infusion by a nurse or other healthcare professional, are different from vaccines, which train the immune system to fight off the virus and to produce antibodies.
“While today’s authorization includes post-exposure prevention of COVID-19 in children, this therapeutic option is not a substitute for vaccination. Vaccines remain our best tool in the fight against the virus and there is a COVID-19 vaccine authorized for children 5 years of age and above,” said FDA’s Cavazzoni.